ALXAN - 8-Ethinylxanthines for the treatment of CNS diseases
Ref.-Nr. 1613
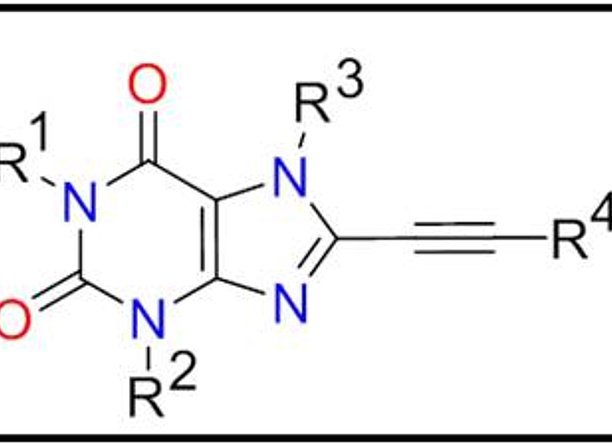
Adenosine is a modulator of many physiological and pathophysiological processes in the central nervous system (CNS). Blockade of the adenosine receptors A1ARs and A2AARs has shown beneficial neuroprotective effects in animal models and in clinical studies of Parkinsons’s disease (PD) and Alzheimer’s disease (AD). Furthermore, selective inhibitors of the monoamine oxidase A (MAO-A) are applied as adjunctive therapeutics for PD, as they protect the brains of PD patients from oxidative stress. Nevertheless, there is still no satisfactory multitarget drug approach which inhibits MAO-A and the two adenosine receptors A1ARs and A2AARs. This invention provides newly designed tricyclic xanthine derivatives which allow overcoming this problem. A variety of 69 derivatives were prepared and evaluated in radioligand binding studies at adenosine receptors and for their ability to inhibit monoamine oxidases. Potent dual-target-directed A1/A2A adenosine receptor antagonists were identified. Several compounds even showed triple-target inhibition. Together, these observations position tricyclic xanthine derivatives as ideal candidates for the treatment of neurodegenerative diseases such as PD and AD, but might also be suitable for the treatment of depression, addiction and restless legs syndrome.
Vorteile
- New potent dual- and triple-target drugs for the treatment of PD and AD
- Multi-target drug with improved compliance, less side effects, reduced toxicity
- Significant advantages over combination therapy: more predictable pharmacology, reduced drug-drug interactions, less complex pharmacokinetics
Kommerzielle Anwendung
On behalf of the University of Bonn, PROvendis offers access to rights for commercial use as well as the opportunity for further co-development.
Aktueller Stand
A European and a US patent have already been granted, other applications are pending.
Relevante Veröffentlichungen
Brunschweiger, A. et al. (2014) 8-Benzyltetrahydropyrazino[2,1-f]purinediones: Water- Soluble Tricyclic Xanthine Derivatives as Multitarget Drugs for Neurodegenerative Diseases. J Med Chem 9;1-22
Koch, P. et al. (2013) 1,3-Dialkyl-substituted tetrahydropyrimido[1,2-f]purine-2,4-diones as multiple target drugs for the potential treatment of neurodegenerative diseases. Bioorg Med Chem 21, 7435-7452
Mertens, M. D. et al. (2014) Alkynyl-coumarinyl ethers as MAO-B inhibitors. Bioorg Med Chem 22; 1916-1928
—
Eine Erfindung der Uni Bonn.