Enantiomerically pure chiral N-acyl-a-aminonitriles - Method for chemical production avoiding the need of toxic reagents
Ref.-Nr. 4654
Keywords: N-acyl-a-aminonitriles, Vildagliptin®, Saxagliptin®, Diabetes, Diabetes type II, Synthesis, gliptins, toxic reagents, pharmaceutical industry, pharma, chemical production, Invention
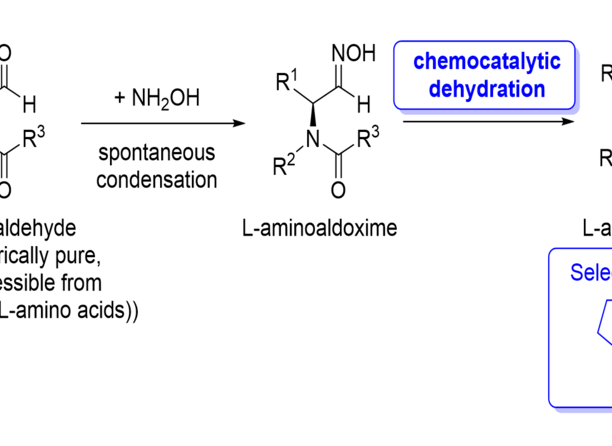
Enantiomerically pure N-acyl-a-aminonitriles gained these days interest in the pharmaceutical industry since a range of recently developed pharmaceuticals are based on this product class. Prominent examples are Vildagliptin®, Saxagliptin® or NVP-DPP-728, which are active pharmaceutical ingredients against diabetes type II and at least two of them are successful already on the market. Important precursors of these so-called “gliptins” are synthesized via multiple phase reactions as state of the art and typical strategies towards such types of molecules are based on toxic reagents such as cyanides or Vilsmeier reagents. Furthermore, the production of the latter one includes toxic precursors like oxalylchloride and phosphorylchloride. Currently, e.g. the synthesis of Vildagliptin® starts from an enantiomerically pure amino acid (here L-proline), which is transformed first into an amide derivative and then via Vilsmeier reagent into the nitrile product (process by Novartis). The present invention focuses on a production process which avoids the need of such toxic reagents. In addition, it has the advantage that the reaction can be conducted under mild conditions. The innovative reaction is also using amino acids as precursors but they are then transformed via an aldehyde intermediate into its corresponding aldoxime, which subsequently is transformed via a chemocatalytic dehydration to the nitrile target molecule.
Vorteile
- Effective chemical production process of N-acyl-a-aminonitriles or N-sulfonyl-a-aminonitriles
- Enantiomerically pure Gliptin derivatives
- New optimized chemocatalytic route
- New approach for important precursors, e.g., of Vildagliptin® or Saxagliptin®
- Starting material easy accessible and avoiding cyanide or Vilsmeier reagents as toxic reagents
Kommerzielle Anwendung
The described technology enables a safe, fast and competitive chemical production of specific N-acyl-a-aminonitriles or N-sulfonyl-a-aminonitriles, serving as intermediates for the synthesis of, e.g., Vildagliptin®. On behalf of the Bielefeld University, PROvendis offers access to rights for commercial use as well as the opportunity for further co-development.
Aktueller Stand
In case of interest we are pleased to inform you about the current patent status.
Relevante Veröffentlichungen
In preparation.
—
Eine Erfindung der Uni Bielefeld.