Anti-tuberculosis drug - PPI-benzimidazole analogues for the treatment of multidrug-resistant tuberculosis
Ref.-Nr. 5126
Keywords: small molecule, tuberculosis, multidrug-resistant tuberculosis, Lansoprazole-sulfide
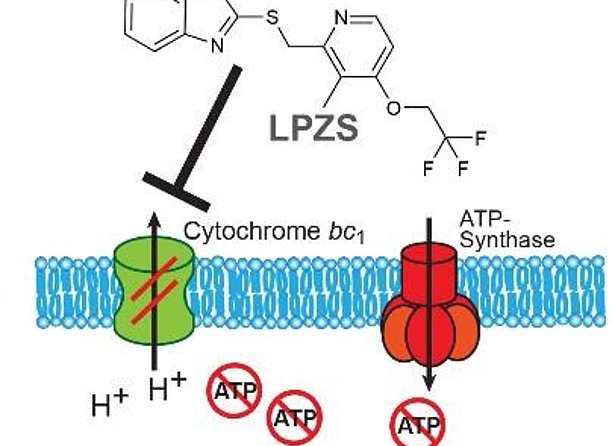
Tuberculosis resulting from infection with Mycobacterium tuberculosis (Mtb) is a serious global health problem accounting for 1.7 million deaths in 2016. A major reason for the high morbidity and mortality caused by Mtb is the long duration of therapy and increasing multidrug-resistance of Mtb strains. Mtb has been treated with combination therapy for over fifty years. Yet according to the latest WHO statistics, approximately half a million new cases of multidrug-resistant tuberculosis are diagnosed every year. Of these, it is estimated that approximately 40,000 have extensively drug-resistant tuberculosis. In view of these numbers, there is a medical need for new anti-Mtb compounds to overcome resistance to known drugs in patients. The inventors were able to demonstrate that lansoprazole and derivatives thereof exhibit growth inhibiting properties against intracellular Mtb. In particular, it has been proven that the reduced form lansoprazole sulfide is the active moiety. Yet, lansoprazole is not metabolized in a sufficient amount to serve as a prodrug. Thus, the inventors propose lansoprazole sulfide (LPZS) as a new drug to treat Mtb infections.
Vorteile
- Active against multidrug-resistant tuberculosis
- Small molecule
- Tested in-vitro and in-vivo
- SAR data available
Kommerzielle Anwendung
LPZS is a promising lead-compound which is distinct from substances identified in other TB-drug screens since its backbone structure has been an approved over-the-counter drug for the past 30 years. Thus, the finding may benefit from 40 years of intense research on pharmacological properties of PPI-benzimidazole analogues and an extensive production pipeline maintained by several pharmaceutical companies. On behalf of University of Cologne, PROvendis offers access to rights for commercial use as well as the opportunity for further co-development.
Aktueller Stand
Patents have been filed for EP and USA based on WO 2016/120259 A1. Efficacy has been proven in-vitro and in a mouse model of Mtb infection. Preliminary SAR data are available.
Relevante Veröffentlichungen
Rybniker, J. et al. (2015) Lansoprazole is an antituberculous prodrug targeting cytochrome bc1. Nat Commun. 6:7659.
Subtil, F. et al. (2018) Activity of 2-(quinolin-4-yloxy)acetamides in Mycobacterium tuberculosis clinical isolates and identification of their molecular target by whole-genome sequencing. Int. J. Antimicrob. Agents 51(3 ): 378–384.
—
Eine Erfindung der Uni Köln.